How to measure depth using a digital caliper |
||||
 |
||||
 |
Depth can be measured by inserting the depth rod into the hole you are measuring. When you adjust the jaws using the thumb screw, the rod will protrude from the end of the caliper. | |||
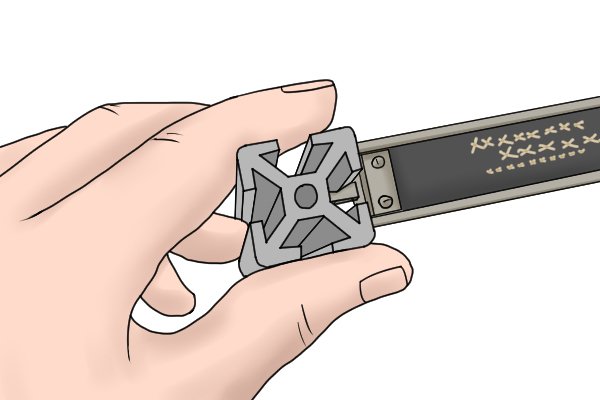 |
Step 1 – Position rodPosition the rod so that it is flat against both the bottom of the hole and an internal side of the hole. Step 2 – Extend depth rod using thumb screwMove the thumb screw gradually until the end of the end of the beam scale makes contact with the top of the hole. |
|||
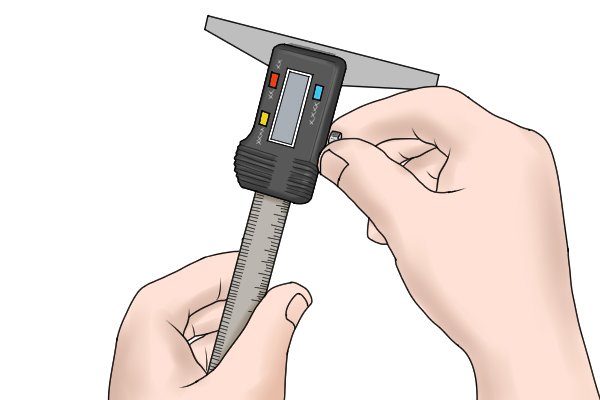 |
Step 3 – Turn lock screwTurn the lock screw to tighten the jaws so that you can remove your object and take your reading. Step 4 – Read measured valueRead the measured value from the LCD display of your digital caliper. |
|||






